In the high-stakes world of pharmaceutical R&D, thousands of drug candidates are abandoned every year long before reaching patients. The harsh reality: fewer than 10% of drugs entering clinical trials achieve regulatory approval, and only a rare few ever become commercial successes. While the industry celebrates its winners, early-stage failures—often the result of avoidable strategic missteps—receive far less attention.
Drug development begins as a survival game. Before committing substantial capital, time, and scientific effort, organizations must answer five foundational questions with clarity and conviction.
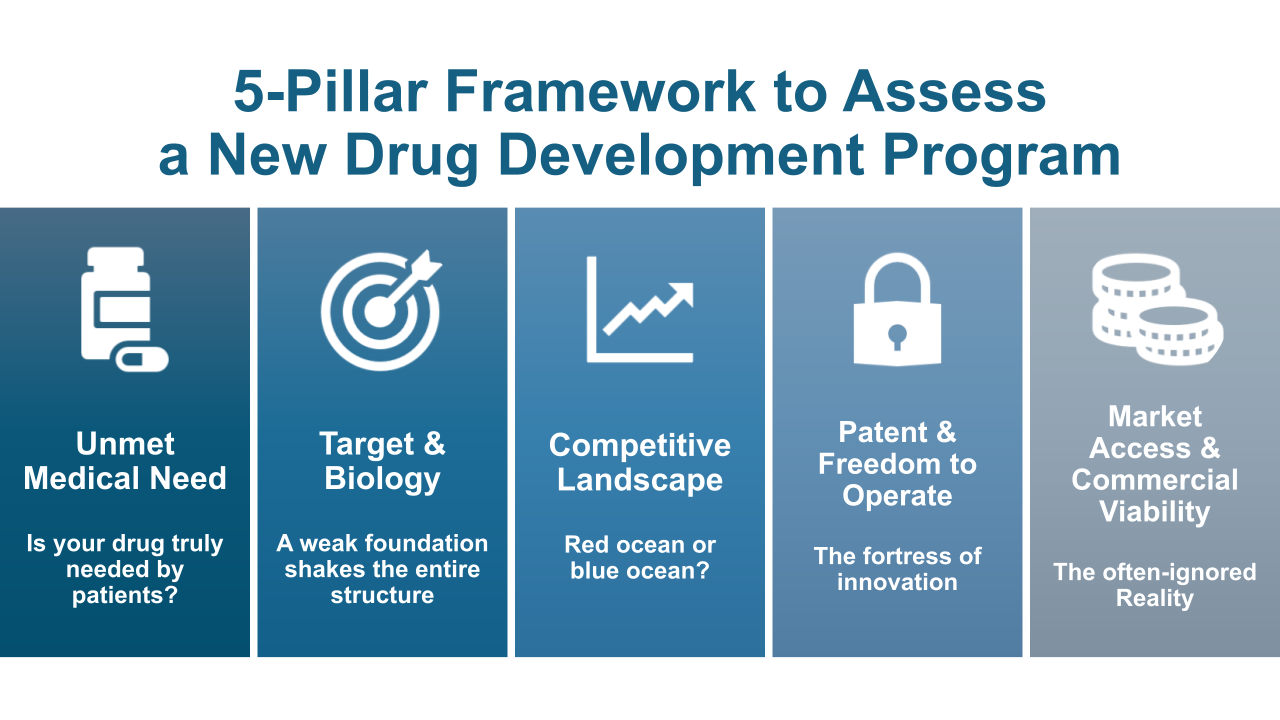
1. Clinical Need: Is Your Drug Truly Needed by Patients?
In many early programs, scientists start with an exciting asset and then search for an indication to match. But without a demonstrable unmet need and a clear clinical value proposition, even promising molecules stall.
Here are the key questions worth examining:
- Unmet Medical Need: Does the target disease lack effective treatments? Where do current therapies fall short—poor efficacy, toxicity, durability, convenience, or resistance? What pain points matter most to clinicians and patients?
- Patient Population Depth: Is the epidemiology solid and well-segmented? How large is the treatable population, and are there biomarker-defined subgroups?
- Clinical Value Proposition: Will your drug, once approved, demonstrably outperform the standard of care—in efficacy, safety, convenience, or cost? Are the improvements meaningful enough to change clinical practice?
A common pitfall is developing “technology for technology’s sake.” For instance, earlier this year, Pfizer discontinued Beqvez, a gene therapy for hemophilia, due to “limited interest” from patients and doctors since its FDA approval in April 2024.
2. Target and Biology: A Weak Scientific Foundation Shakes the Entire Structure
A drug is only as strong as its biological rationale. Weak or incomplete target validation remains a leading cause of attrition.
The core components to review are:
- Target Validation: Is there a robust chain of evidence—from human genetics (e.g., GWAS), clinical samples, and preclinical models—supporting the target–disease relationship?
- Mechanism of Action: Is the drug’s mechanism well understood, and does it meaningfully influence the disease pathway?
- Biological Risk Assessment: Does the target sit within a complex network? Are there known off-target risks, compensatory pathways, or resistance mechanisms?
The case below illustrates how this can play out: Years of investment in β-amyloid antibodies for Alzheimer’s disease have faced repeated Phase III failures. A central scientific debate persists: does clearing amyloid plaques truly alter the core disease process?
3. Competitive Landscape and Differentiation: Red Ocean or Blue Ocean?
The biopharmaceutical landscape often behaves like the fashion industry. Not long ago, headlines were dominated by PD-1 inhibitors; then came the ADC wave; now the GLP-1 era is in full swing.
Following trends is not inherently wrong—but entering a crowded field without a differentiation strategy is risky. Some of the essential elements to consider are:
- Competitive Mapping: Identify all marketed and pipeline competitors, especially those in late-stage trials. These are the benchmarks your drug will face upon launch.
- Differentiation Strategy: Can your molecule achieve:
- Superior efficacy?
- A better safety profile (e.g., reduced cardiotoxicity, fewer immune-related events)?
- Improved convenience (oral vs. injectable, long-acting vs. daily dosing)?
- Broader or more resistant patient coverage?
- Market Timing: Will your program be first-in-class, best-in-class, me-better, or a me-too commodity?
One clear warning sign is when your drug merely matches a Phase III competitor with no clear advantage. This substantially increases both commercial and development risk.
4. Patent and Freedom to Operate: The Fortress of Innovation
Strong IP protection is often underestimated by early-stage teams, yet it determines whether a molecule has defensible and monetizable value.
The critical IP issues to evaluate are:
- Core Patent Strength: Are your composition, polymorph, and method-of-use patents broad, defensible, and filed in key jurisdictions?
- Freedom to Operate: Have you conducted comprehensive searches to ensure your R&D, manufacturing, and commercialization plans do not infringe on others’ patents? Pay special attention to enabling technologies such as gene editing, vectors, or delivery platforms.
- Lifecycle Management: Consider follow-on patents (e.g., formulations, combinations, new indications) early to extend exclusivity.
Here is a hard lesson that underscores the consequences of overlooking IP risks: A biotech company once discovered—near IND filing—that its gene-editing method infringed a foundational IP family. The program was halted, and the company’s valuation dropped sharply.
5. Market Access and Commercial Viability: The Often-Ignored Reality
Many scientists assume market access is a concern for later stages or for large pharma partners. In reality, commercial viability shapes investor decisions and valuation from day one.
A thorough commercial evaluation should include:
- Pricing Ceiling: What reimbursement systems (public, private, self-pay) apply? What are the pricing benchmarks for similar therapies? Can payers justify your anticipated price?
- Peak Sales Forecast: Build conservative, base-case, and optimistic scenarios using patient numbers, pricing, penetration, and duration. Will projected revenues support total development costs?
- Budget and Funding Strategy: Account for all costs from program initiation through launch—including the hidden costs of failure. Can your current or expected funding carry the program to value-inflection milestones?
It’s important to recognize that even a scientifically sound therapy may struggle if the population is ultra-rare and pricing cannot exceed payer thresholds. This can make fundraising and commercialization unsustainable.
Final Thoughts
At Polygon Health Analytics, we believe successful drug development begins with foresight and thrives on strategic precision. Before advancing a drug candidate, ensure you have clarity on these five critical pillars:
- A meaningful and sizable clinical need
- A validated and compelling biological mechanism
- Clear and defensible competitive differentiation
- Robust patent protection and freedom to operate
- A realistic and attractive commercial outlook
Strategic clarity at the starting line can make the difference between a breakthrough and a breakdown.